The nature of research is such that new discoveries build on previous research. For this process to continue, it is often necessary to share research materials and data with other researchers. The way institutions share research materials and data is often by way of material transfer agreements (MTAs), Data Use Agreements (DUAs), or Research Collaboration Agreements (RCAs).
A DUA or DTUA is a contract that dictates the terms of transfer of nonpublic data from one party (a data provider) to another (a data recipient). The purpose of the DUA is to protect the sensitive data and intellectual property rights, as well as other rights, of the provider, as well as the data subject, while permitting intended research use with the data.
Variables of Data Transfers
The type of data being shared and the factors involved in the data sharing can vary greatly from one project to another. Most often, DUAs are capturing the protections and intended uses of transferring protected health information (PHI) in the form of a Limited Data Set as defined by HIPAA.
Other times, DUAs may contemplate the sharing and use of:
- Human patient data which is fully de-identified
- Personally identifiable information (PII)
- Family Educational Rights and Privacy Act (FERPA)
- Federal or state-protected human data
- Non-human data (i.e. companion animals)
- Other nonpublic data or intangible material
Is a DUA needed?
Due to the varying nature and complexities of any one project from another, SPA is able to offer assistance and resources for your determination of whether or not a DUA or other data sharing agreement is necessary. However, SPA cannot make this determination for you.
Please see the helpful guidance documents here for assistance with determining whether or not your data can be shared, whether or not a data agreement may be needed, and what type of data terms or document(s) may be necessary:
- Exhibit A: Can the data be shared?
- Exhibit B: Is an agreement needed?
- FDP’s guidance on data agreements
DUA requests
All DUAs, DTUAs, data license agreements and the like must be reviewed by the office of Sponsored Projects Administration (SPA).
These steps explain the process of completing an incoming MTA request:
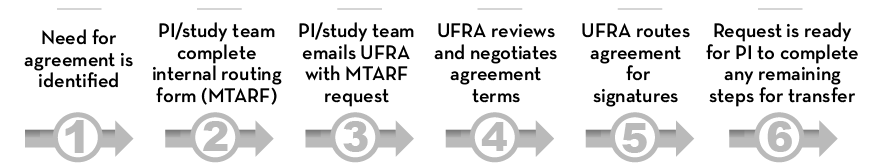
- When you have identified the need for a data sharing agreement, create a Material Transfer Agreement Routing Form (MTARF) (see #2 below), and then send an email to ufra@umn.edu (the master email of SPA's Unfunded Research Agreement (UFRA) staff). Please list the UMN Principal Investigator name and the MTARF number in the subject line of your email. Please also include a brief description of the planned research, whether in the body of the email or as an attachment, so that UFRA staff can best tailor the DUA terms to protect the research.
- To create an MTARF, go to the EGMS homepage, select "EGMS forms," log in, and select "Make request," then "MTARF." See MTARF instructions or view a completed sample form.
- Once the MTARF is completed and approved by the principal investigator, email ufra@umn.edu and include the MTARF number, PI name, and name of the other entity in the subject line. The MTARF system currently does not notify our team of these forms and we rely on your email to provide us with this notice.
Note: If the entity has provided a draft DUA agreement, include the attachment in your email.
In most cases, the UFRA staff will need additional information pertaining to your data agreement needs. Please also complete the questions in the Data Agreement Request Questions form which will be routed to the team automatically following your completion. UFRA staff will pair these responses with your MTARF form.
- The UFRA staff will review your request and contact you if more information is required. You may track the progress of your request by searching the UFRA database for your agreement by PI name, the other party, or MTARF number.
The UFRA staff will review the terms of the MTA to see how well they protect the interests of the PI and the University. They will then negotiate directly with the other party to reach agreeable terms within the DUA ahead of finalizing and executing the DUA. The time required depends on the willingness of the other party to negotiate and the timeliness of their actions as well as each party’s workload levels at the time. If this is an urgent request, be sure to advise the UFRA staff of this in your email (see Step 3 above).
- Once both parties have agreed to the terms of the MTA, the MTA will be sent to the material provider for signature and a copy will be sent to you.
Note: A Principal Investigator cannot sign the DUA on behalf of UMN. SPA will sign to legally bind UMN to the DUA terms.
- You will receive a copy of the fully signed DUA from the UFRA staff once it is available.
Questions?
We're Here to Help! Email ufra@umn.edu